Oxford Nanopore (ON) has been developing a disruptive nanopore-based technology for sequencing DNA, RNA, proteins, and other long-chain molecules since its birth in 2005. The company has just announced that within the next 6-9 months it will bring to market a fast, portable, and disposable protein sequencer that will democratize sequencing by eliminating large capital costs associated with equipment required to enter the field.
The MinION is the size of a USB memory stick, and obtains both power and computer analysis from a normal laptop computer. No polymerase chain reaction (PCR) or other DNA amplification technique is needed for optimum sensitivity. The MinION is capable of sequencing 100 million base pairs within its six hour working life. Samples of whole blood, plasma, and serum are accepted by the device for immediate analysis and the reagents needed for the analytic process come ready to use with each MinION.
What are nanopores?

A nanopore is an organic molecule penetrated by a very small (~1-2 nanometer) hole. These nanopores are embedded in a synthetic polymer membrane which is only two molecular layers in thickness. Initially bilayer lipid membranes, similar to those appearing throughout the body, were used, but proved far too fragile to use in a production sequencer. Development of such a thin membrane that has extremely large electrical resistance and is sufficiently rugged for use in the field was a major (and successful) ON project.
A nanopore is mounted upon such a membrane so that the hole in the nanopore forms a path from one side of the membrane to the other. The membrane is then positioned so that it divides a volume of an electrophysiological fluid in half.
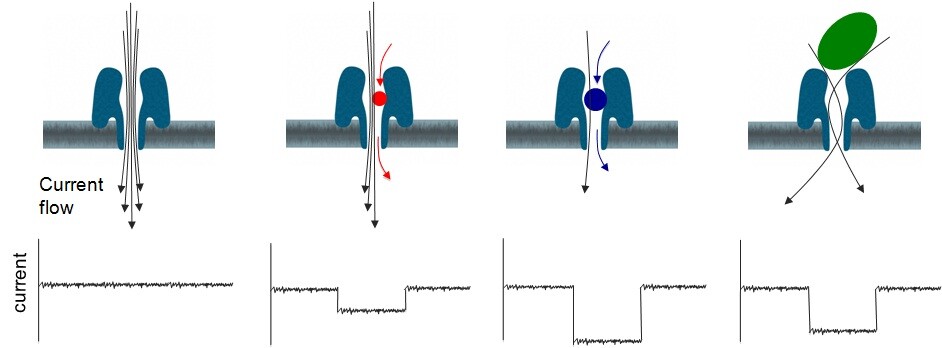
The image above shows a nanopore embedded in a membrane. A voltage difference is placed between the two halves of the fluid. The fluid contains mobile ions, so an ion current then passes through the center of the nanopore. This ionic current acts to sweep nearby molecules, proteins, and even DNA or RNA into the mouth of the nanopore.
If a target molecule is drawn into or upon the nanopore, it disrupts the flow of ions in a characteristic manner which can be detected and interpreted.
Although in the image it appears that the detection current is only due to physical obstruction of the nanopore, one must remember that the chemical bonds of the nanopore and the target molecules have unique distributions of electronic charge. The target molecule will attempt to lodge in the nanopore so that the potential energy of the system is lowest. This effect, together with the possibility of modifying the structure of the nanopore to increase the difference in the ionic current for a set of target molecules implies that selectivity can be adjusted to detect small differences in molecular structure.
A wide range of molecules can be detected and identified using the nanopore ionic current measurements, ranging from small physiological or pharmaceutical molecules to proteins. However, the MinION is directed toward DNA sequencing, so the nanopore and enzymes in the electrophysiological fluid are optimized to differentiate the four amino acids (adenine, cytosine, guanine, and thymine) which encode genes in DNA. In the final result, three amino acids are read simultaneously by the nanopore, which possesses the delicacy to differentiate between the 64 possible combinations.
Strand sequencing
The MinION uses a strand sequencing technique for DNA analysis. Strand sequencing is shown in the following excellent video from Oxford Nanopore.
Here's a quick overview: The DNA strand is attracted to an enzyme that is part of the electrophysiological fluid. In turn, the enzyme is attracted to a nanopore, in which it lodges. The enzyme is engineered to unzip the double-helix structure of the DNA strand so that one strand is directed back into the fluid, while the other enters the nanopore for sequencing. More amazing is that the enzyme incorporates a ratcheting structure, so the single DNA strand does not pass quickly through the nanopore, but rather advances one base pair at a time. The rate at which the DNA strand passes through the nanohole is controlled by the magnitude of the unobstructed ionic current, and can be as large as 15 base pairs per second. Better accuracy is associated with slower rates.
The sensing electronics for the ionic current is fabricated on a sample chip, which comprises 512 nanopores, giving a total strand reading rate of about 7500 bases/second. The MinION has a limited operational life of about 6 hours, but in that time can read over 150 million bases.
For comparison, the largest human chromosome, chromosome 1, contains about 250 million base pairs. Attempting to read a smaller complete chromosome (say, number 22, which has about 50 million base pairs) would require that the chromosome be split into a large number of pieces which would be analyzed concurrently, and the pieces reassembled. This is not the primary purpose of MinION, but it does provide data which would make possible such a feat.
Bringing genetic analysis out of the research lab
The MinION has successfully been used to read the genome of the lambda bacteriophage, about 48500 base pairs in length, twice in a single pass. This was accomplished by closing the end of the DNA with a hairpin closure, so that both strands in the genome would pass through the same nanopore. Reading roughly 100,000 base pairs in a single DNA capture is unheard of using more traditional sequencing techniques.During interviews just proceeding the product announcement at AGBT 2012, Clive Brown, the Chief Technology Officer of Oxford Nanopores, revealed that the expected $900 price tag for the MinION has a good bit of margin built in. We can thus expect prices to fall quickly as production becomes routine in its challenges.
The science and engineering research which has gone into making a device like the MinION possible is a remarkable feat for the ages. It is difficult to point to any portion of the development which was straightforward. It is to be hoped that the impact of MinION and future developments thereof is to truly begin to bring genetic analysis out of the research lab as a vital tool for physicians' office.
Source: Oxford Nanopore











