Among the primary causes of adult-onset blindness are degenerative diseases of the retina, such as macular degeneration and retinitis pigmentosa. While some treatments have been developed that slow down the rate of degeneration, the clinical situation is still generally unsatisfactory. But if you could grow a new retina, transplant might be a possible cure. Now new hope is springing up from a research project at the University of Wisconsin-Madison in which scientists have succeeded in growing human retinal tissue from stem cells.
Pluripotent stem cells are capable of forming nearly any tissue in the body including retinal tissue. There has been great controversy about using pluripotent stem cells for human research or treatment, as historically the only source was to harvest them from early stage human embryos. Instead, for this work the researchers were able to regress mature body cells back into the pluripotent stem cells from which they originally grew. The process is called reprogramming, and is accomplished by inserting a set of proteins into the cell.
To produce the pluripotent stem cells, a white blood cell was taken from a simple blood sample. Genes which code for the reprogramming proteins are inserted into a plasmid, a nonliving ring of DNA. The cell is then infected with the plasmid, rather as a virus infects a cell, with the difference that the plasmid's genes do not become part of the cell's genetic structure. As the reprogramming proteins are formed within the cell by the plasmid DNA, the cell has a good chance of being reprogrammed into a pluripotent stem cell. This stem cell can then be encouraged to grow and differentiate into retinal tissue rather than make more blood cells.
Laboratory-grown human retinal tissue will certainly be used in testing drugs and to study degenerative diseases of the retina, and may eventually make available a new transplantable retina, or a new retina that is grown in place within the eye.
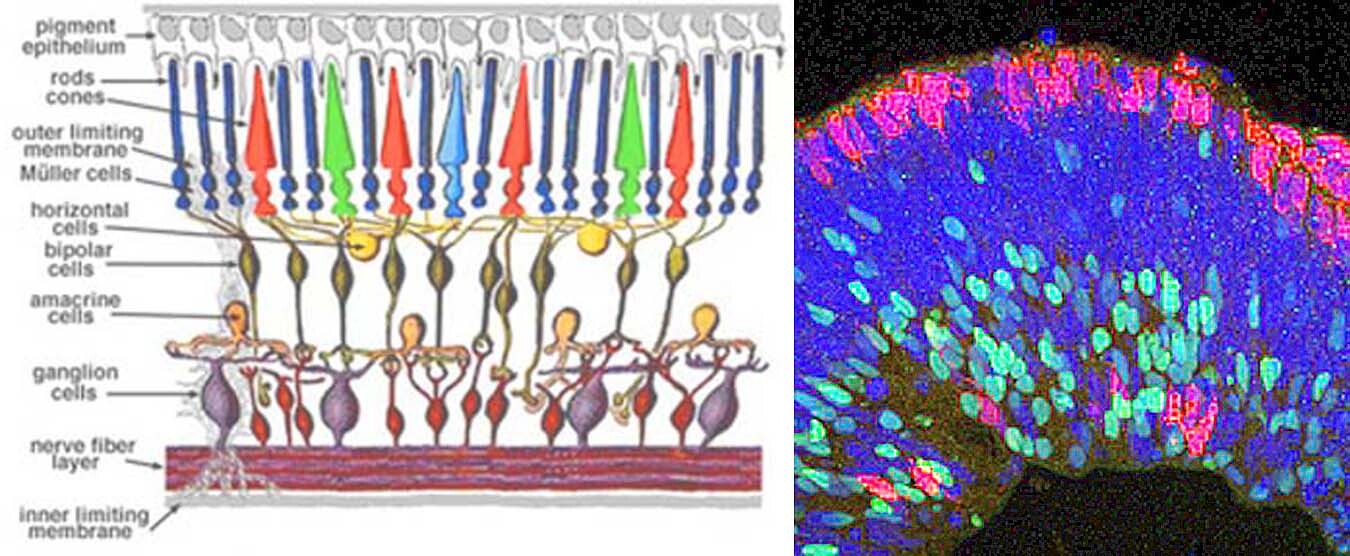
The figure above compares a schematic of the human retina with a photomicrograph of laboratory-grown retinal tissue. The new tissue has separated into at least three layers of cells, with rudimentary photosensitive rods or cones (red) at the top of the picture, and nerve ganglia (blue-green) at the bottom. The blue cells in the middle layer are likely bipolar retinal cells. The structure of the lab-grown retinal tissue is similar to that of a normal human eye, as can be seen by comparison with the retina schematic. The cells also formed synapses, which provide the channels through which optical information flows to the brain.
"We don't know how far this technology will take us, but the fact that we are able to grow a rudimentary retina structure from a patient's blood cells is encouraging, not only because it confirms our earlier work using human skin cells, but also because blood as a starting source is convenient to obtain," says Dr. David Gamm, pediatric ophthalmologist and senior author of the study. "This is a solid step forward." Further steps are eagerly awaited by those living in the dark.
Source: University of Wisconsin School of Medicine and Public Health






