Researchers at MIT have come up with a new design for a rechargeable flow battery that does away with the expensive and ineffective membrane of previous designs. The device could prove the ideal solution for effectively storing energy from intermittent power sources such as solar and wind power.
What is a flow battery?
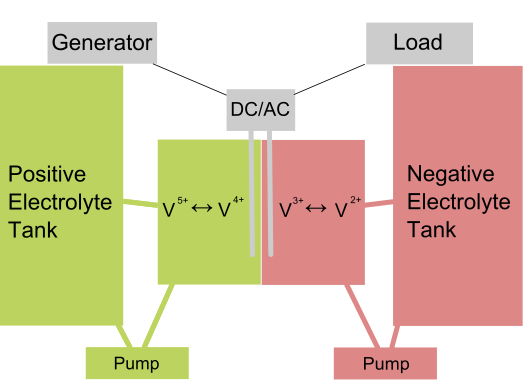
A flow battery is a special type of rechargeable battery in which two liquids with opposite electric charge (electrolytes) exchange ions, converting chemical energy directly into electricity. The electrolytes are usually separated by a thin membrane that lets them exchange ions without mixing.
The electrolytes are stored separately from the cell itself, in two big tanks, and the electrolyte is pumped into the cell as needed. This means the system can be scaled easily, simply by changing the size of the tank. Doing so can produce systems of vastly different capabilities, from a few kWh up to several MWh.
Scalability aside, flow batteries come with many more perks: they can stay idle for long periods of time without losing their charge, they have a quick response time, and they can charge and discharge quickly just by replacing the electrolyte fluid. For these reasons, over the past few years some people have advanced them as a way to quickly refuel electric cars.
On the flipside, flow batteries are more complicated than standard batteries, each requiring their own system of pumps and sensors; moreover, energy densities are usually lower than those of your standard Li-ion battery.
A battery for the future
The main challenge in developing an effective flow battery has been to find a good balance between performance and costs. The electrolytes used are typically not very expensive, but they tend to eat away at the costly membrane, shortening the battery's lifetime. The MIT team's solution circumvents the issue in perhaps the most elegant of ways – by taking out the membrane altogether.
The small flow battery prototype built by the researchers uses a curious phenomenon in fluid dynamics called laminar flow: if both liquids are kept at low enough speeds and other conditions are satisfied, the two electrolytes won't mix, making the membrane superfluous.
Pumping liquid bromine over a graphite cathod and hydrobromic acid and hydrogen gas over a porous anode, the researchers created a reservoir of free electrons that can be discharged or released at will.
While other teams had attempted a membraneless design before, this is the first one in which the battery can be recharged as well as discharged. Their flow battery produced up to 0.795 watts per square centimeter: that's three times as much as other membraneless systems, and about 10 times higher than most lithium-ion batteries.
Future developments
The researchers also generalized the manufacturing parameters of the flow battery, by creating a mathematical model that they can use to optimize the system and eventually build larger-scale devices that are better suited to grid applications.
Previous membraneless systems have been largely unpractical, but the scaled-up version of the device could have a substantial real-word impact because it could be used to produce energy for a very competitive US$100 per kilowatt-hour. "Most systems are easily an order of magnitude higher, and no one’s ever built anything at that price," says William Braff, who was part of the research team.
One area in which this technology could be put to good use is the storage of renewable energy. Since sunlight and wind are highly unpredictable power sources in the short term, being able to store large quantities of clean energy to use as a buffer is essential if green energy is going to continue to satisfy a larger and larger portion of our energy needs.
The team's research appears in the journal Nature Communications.
Sources: MIT, Electropaedia





