Twenty to thirty percent of the world's population suffers from some sort of chronic pain, which is far more difficult to control than, say, the pain of a cut or bruise. A great deal of effort has gone into the search for medically acceptable ways to control such pain, with few good answers emerging. Now medical researchers at the University of Michigan have directly demonstrated that transcranial electrical stimulation of a patient's brain causes the release of a natural opiate that dulls or eliminates the perception of pain.
Transcutaneous electric nerve stimulation (TENS) has been used far back into history to relieve chronic pain, with limited success. More recently, however, transcutaneous direct current stimulation (tDCS) has become an accepted, if experimental, method of producing changes in brain function, with multiple applications in treatment of chronic pain. Requiring little more than a nine-volt battery and a pair of electrodes, it involves passing about 1-2 mA of current through the skull and the underlying brain tissue. One of the most important aspects of tDCS is its ability to maintain cortical changes even after the stimulation is ended.
tDCS has been found to speed recovery of stroke patients, help lift a depressed mood, improve memory, speed up training of military snipers, and even improve one's ability to do mathematics. Prior studies by the Michigan group demonstrated that tDCS can decrease the intensity and pain of chronic migraine patients. However, the mechanism was not completely understood at that time.
There are two main classes of chronic pain: chronic pain due to a physical cause, and chronic pain due to nerve damage. Both types can become entrenched physically into the nervous system and brain through neuroplasticity. Roughly speaking, the brain gets into the habit of perceiving pain, and with practice becomes quite skilled at feeling a wide range of sensory inputs as pain. Chronic pain can result from numerous conditions, including arthritis, spinal stenosis, cancer, fibromyalgia, migraine headaches, trigeminal neuralgia, and nerve damage, among many others. In some cases, nothing is clinically observed save for the pain itself.
In the new Michigan study, positron emission tomography (PET) antibody studies showed that tDCS stimulation of the motor cortex causes the active release of an opiate-like substance (beta-endorphin) considered to be one of the body's most powerful painkillers. The tDCS unit was set to supply two mA of current, with electrons being introduced into the primary motor cortex and removed through the forehead.
The patient suffered from intense chronic pain from trigeminal neuralgia, a disease of the trigeminal nerve that produces episodes of such intense pain that it has been called the "suicide" disease. One 20 minute tDCS treatment significantly reduced the patient's pain reaction to a cold stimulus, but relief of the chronic pain required multiple treatments. This suggests a distinction between acute and chronic pain, wherein chronic pain becomes habitual to the brain through neuroplastic strengthening of the pain perception circuits. This observation helps to explain why chronic pain is so difficult to treat.
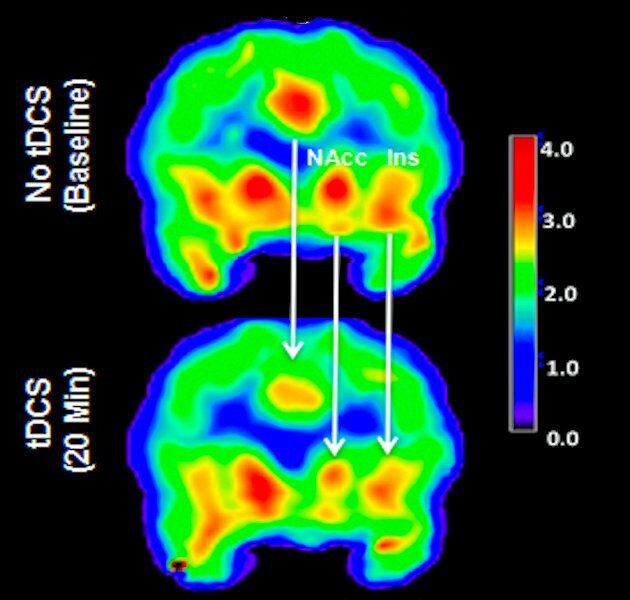
The PET scans above show the brain of a chronic pain patient before and during electrical stimulation to induce pain relief. The areas in red represent the relative number of free mu-opioid receptors, the main target of pharmaceutical opiates. They found a decrease in availability of those receptors following electrical stimulation. The results indicate for the first time the active release of endogenous mu-opioid agonists in response to tDCS treatment.
"This is arguably the main resource in the brain to reduce pain," said Prof. DaSilva, one of the lead researchers. "We're stimulating the release of our (body's) own resources to provide analgesia."
Next, researchers will investigate long-term effects of electric stimulation on the brain and find specific targets in the brain that may be more effective for particular pain conditions.
The team's study appears in the journal Frontiers in Psychiatry.
Source: University of Michigan





