The advent of unmanned combat vehicles is generating a need for smaller weapon systems to fit their reduced dimensions. As a result, more powerful explosives are being sought to get the most performance from smaller warheads. Introduction of new explosives is a rather slow process, as premature detonation of an explosive is extremely embarrassing. The desire for higher-performance explosives persists, though, so explosive chemists get used to dancing along the edge of instability. Fortunately, new chemistry occasionally appears that pushes the edge back a bit. The recent synthesis of a stable, high-performance explosive by a research team at the University of Michigan indicates that such new chemistry is now at hand.
An ideal explosive combines the attributes of high explosive power, high stability, high density, low environmental impact, and low cost. Perhaps a dozen favored explosives, including TNT, RDX, HMX, PETN, TATB, and HNS, dominate current weaponizable explosive formulation. Improving on the favored explosives usually requires improving one attribute without significantly degrading others.
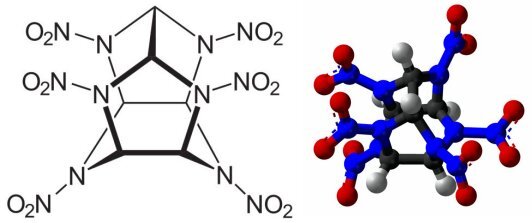
Otherwise known as CL-20, 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane (C6H6N12O12) is a relatively new candidate for inclusion into the group of favored explosives. Originally synthesized in 1986 by Arnold Neilsen at the Naval Surface Weapons Center at China Lake, California (hence the CL- designator), CL-20 is the highest energy compound as well as the highest density compound known among organic chemicals. It is manufactured in the dense episilon crystal phase in batches of about 100 kg (220 lb) by Thiokol and the French SNPE. The cost in kilogram lots is quoted at over US$1300/kg, which would be expected to fall by a factor of perhaps five or ten when production is scaled up to support manufacture of active weapons.

The current state-of-the-art military explosive is HMX at a cost of about US$100/kg. The photo above makes clear that CL-20 is considerably more powerful than HMX, demonstrating about 40 percent deeper penetration in steel blocks. The additional power results from the combination of faster detonation velocity (9,660 m/s compared to 9,100 m/s for HMX) and larger density (2.04 g/cc compared to 1.91 g/cc for HMX).
The increased power of CL-20 argues for its use in smaller weapon systems, such as unmanned air vehicles. However, CL-20 is rather susceptible to impact and friction, being about as sensitive as PETN, the least stable of the common military explosives. Large-scale tests have mostly used a combination of CL-20 and a plastic binder in a 90-10 ratio. While this plastic-bound explosive has achieved a higher level of stability by separating the CL-20 crystals, the power of the explosive is reduced to roughly the HMX level.
The history of CL-20 is somewhat disappointing, but there simply are not that many candidates for new explosives, so people kept experimenting with its use. Then Professor Adam Matzgar of the University of Michigan Chemistry Department set his research team on the problem.
When you can't change the chemicals, you change their environment. Cocrystallization is a method for engineering solid forms of difficult materials that has been quite successful in producing new pharmaceuticals. A normal mixture of two fine powders produces a jumbled heap of the two powders – the immediate neighborhood of each powder is the same as if it were the only powder in the mixture. As a result, explosive properties of such a mixture often lie between the properties of the two pure materials.
In cocrystallization, both materials are crystallized from the same liquid in such a manner that a molecular solid of the two materials is formed. A molecular solid is one in which the structure and order of the two components is relatively fixed. A one-to-one cocrystal of A and B will have alternating molecules of A and B throughout the cocrystal, with the relative orientation and spacing of A and B being fixed as well. This changes the local environment of each of the components in the cocrystal, which also changes its explosive properties.
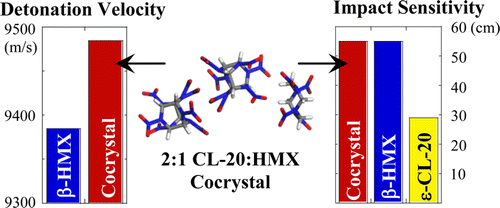
Prof. Metzgar's group succeeded in forming a cocrystal having two molecules of CL-20 to one molecule of HMX. By simple averages one would expect that the detonation velocity would be about 9,470 m/s, and the impact detonation threshold (the distance over which the fall of a standard weight will set off an explosive) would be about 38 cm. The cocrystal did indeed have a detonation velocity of 9,480 m/s, in good agreement with the expected value. However, the impact detonation threshold was 55 cm, essentially identical to that of pure HMX. In the environment of the cocrystal, the stability of the CL-20 molecule is sufficiently enhanced that the HMX becomes the more sensitive component. There is more of a power difference between HMX and the cocrystal than might be immediately apparent, as the cocrystal has larger density than does HMX, leading to a power increase of about 20 percent over pure HMX. To give this number a reference point, the cocrystal is a bigger improvement over HMX than HMX is over RDX.
The unexpected insensitivity of the cocrystal is thought to reflect the increased density of hydrogen bonds in the cocrystal relative to the crystals of pure HMX and CL-20. Intuitively, the instability of a molecule probably has something to do with chemical groups moving relative to the core of the molecule, and additional bonds serve to hold the groups in place. By being more powerful and safer to handle, the cocrystal presented is an attractive candidate to supplant the current military state-of-the-art explosive, HMX.
Most high-explosive anti-tank (HEAT) weapons and their relatives cost far more than their explosive charges. An example is the AGM-114 Hellfire missile, which costs about US$58,000 and has eight or nine kilograms (17 or 19 lbs) of explosive aboard. If a ten percent increase in cost provides substantially better performance, it seems likely that the military would pay the price.
Source: American Chemical Society







