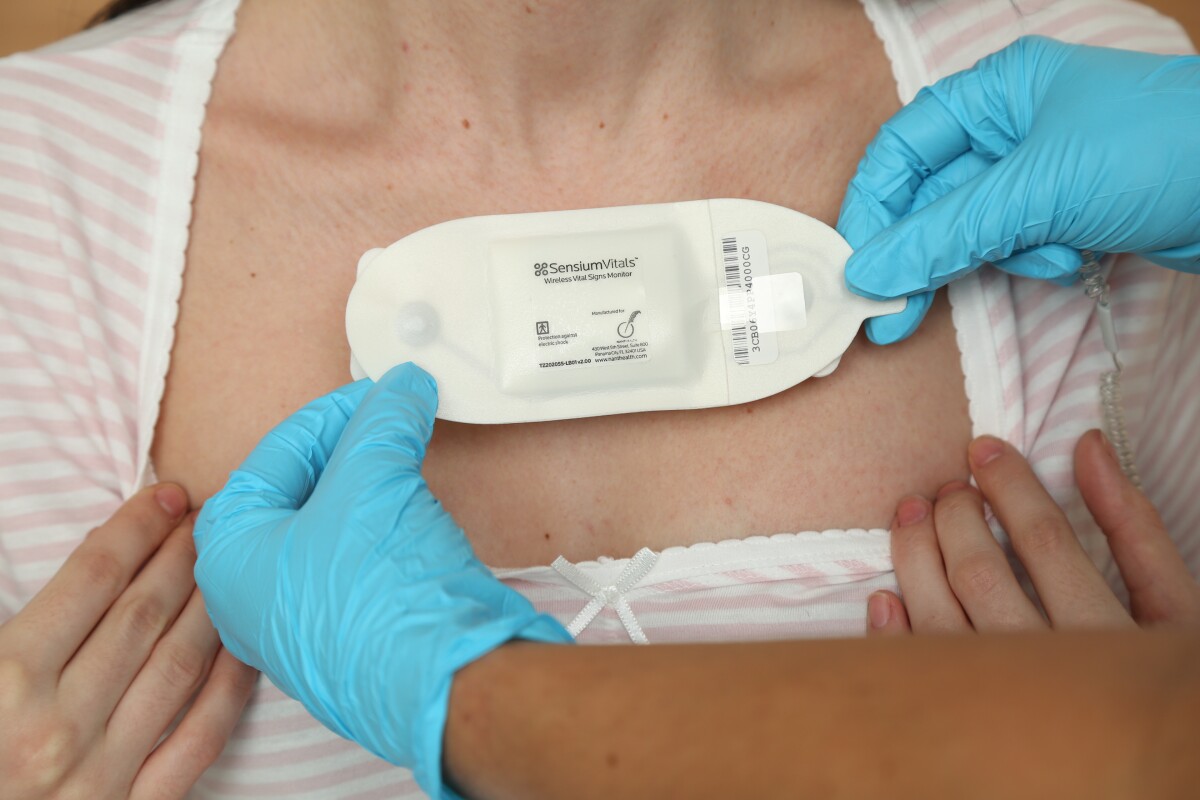
A new system developed by the Toumaz Group may hold the key to improving the level of life-saving attention received by patients, providing two-minute updates on their vital signs 24 hours a day with the use of a comfortable, wireless sensor pad. The equipment, known as SensiumVitals, is claimed to be so portable and convenient that it could even be used in the future to provide hospital-level observation to individuals in the comfort of their own homes.
The SensiumVitals system takes the form of a compact, single-use sensor pad, that when placed on the patient's chest, feeds regular updates on the their vital signs to hospital staff. According to the Toumaz Group, this will allow medical professionals to monitor the well-being of their patients in a more comprehensive and efficient manner. The system should also have the effect of creating a more relaxed atmosphere for nurses working a ward, allowing them to be more focused on the patient at hand, safe in the knowledge that their other patients are under near-constant observation.
Under current practices, vital signs are monitored roughly once every four to eight hours, leaving a significant period of time for the patient to deteriorate between checks. This deterioration can lead to severe complications that could either result in the death of the patient, or at least the need for further treatment, heightening the cost of an illness. Increased observation could cut down on these ordinarily unavoidable complications, however it is worth noting that SensiumVitals will not replace current routine checks, but is rather designed to bolster them.
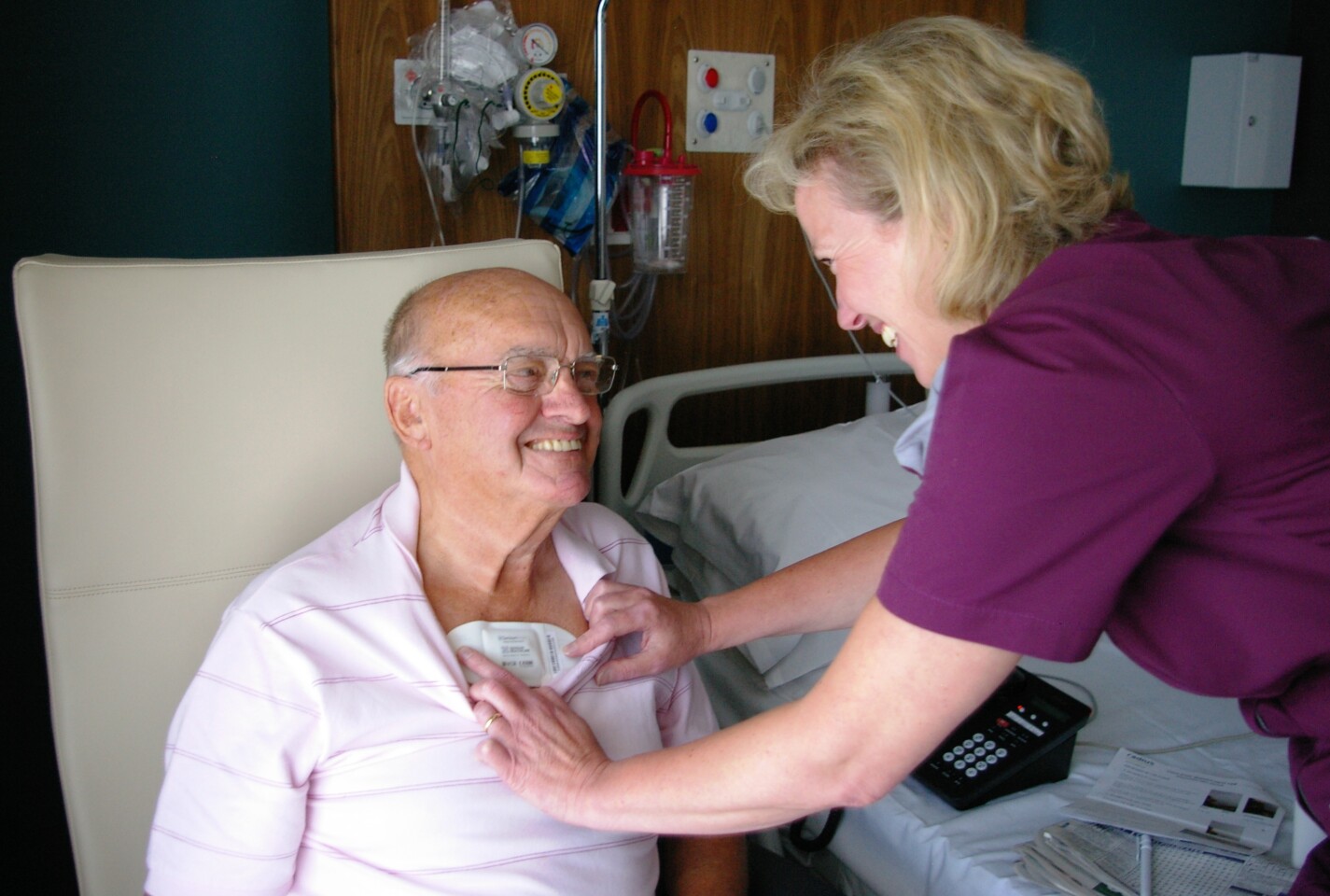
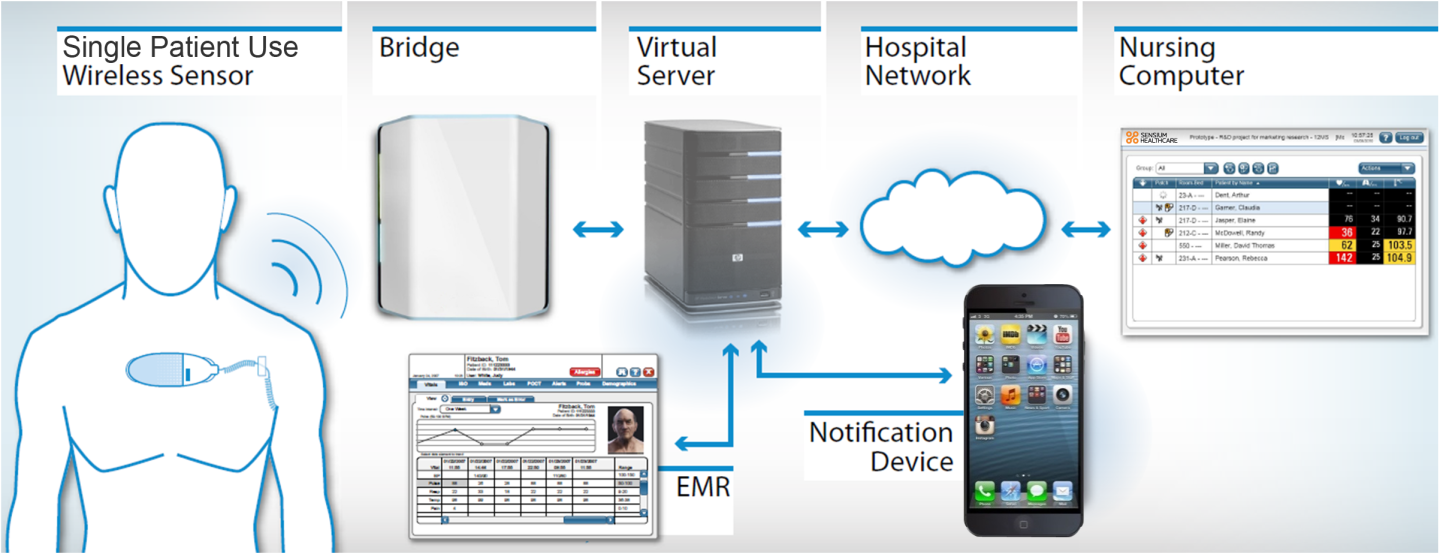
The sensors attach to the chest of the patient using ECG pre-gelled electrodes and are reportedly so non-invasive and comfortable, that patients who have trialed the equipment often forget that they are wearing it. The pad has a five-day battery life, and makes use of a new radio chip that allows for wireless communication between the SensiumVitals equipment, and the hospital's own IT infrastructure via a wall-mounted bridge.
The Toumaz Group has obtained impressive results in the past with its wearable health monitors, but to successfully apply similar technology in a hospital environment required a more versatile approach to data distribution. By linking with the hospital's system, SensiumVitals can push notifications to nursing computers, pagers, or even to the mobile phones of hospital staff, alerting them should any anomaly in patient heart rate, breathing, or temperature be detected.
Doctors and nurses can tailor these notifications to individual patients, setting the parameters for an alert to take into account the patient's age, gender, condition and even previous baseline vital signs if available. This has the combined effect of making the alert system more sensitive to an individual patient's ailment, while also cutting down on nuisance notifications that may arise from incorrect baseline data.
SensiumVitals has already received FDA approval, and in October 2012 underwent a six-month case study at Saint John’s Health Center, Los Angeles. The results of the study were overwhelmingly positive, with the pilot detecting and facilitating early intervention in 12 percent of the 168 patients involved in the trial.
Furthermore the system was shown to be economically viable, as the device led to shorter hospital stays due to the reduced number of complications, potentially saving the ward involved in the trial an estimated US$200,000 per year.
More recently the SensiumVitals system has been trialed in the Montefiore Hospital, Brighton, UK. The system has been in place since late May, and has already received high acclaim from staff and patients alike.
"We are very encouraged by the feedback we have received from the Montefiore Hospital in Brighton" states Anthony Sethill, CEO of the Toumaz Group. "In the coming months, we expect to introduce the system into a significant number of hospitals both in the private sector and across the NHS."
The video below highlights the benefits of the SensiumVitals system observed during the trial at Saint John’s Health Center, Los Angeles.
Source: Toumaz Group