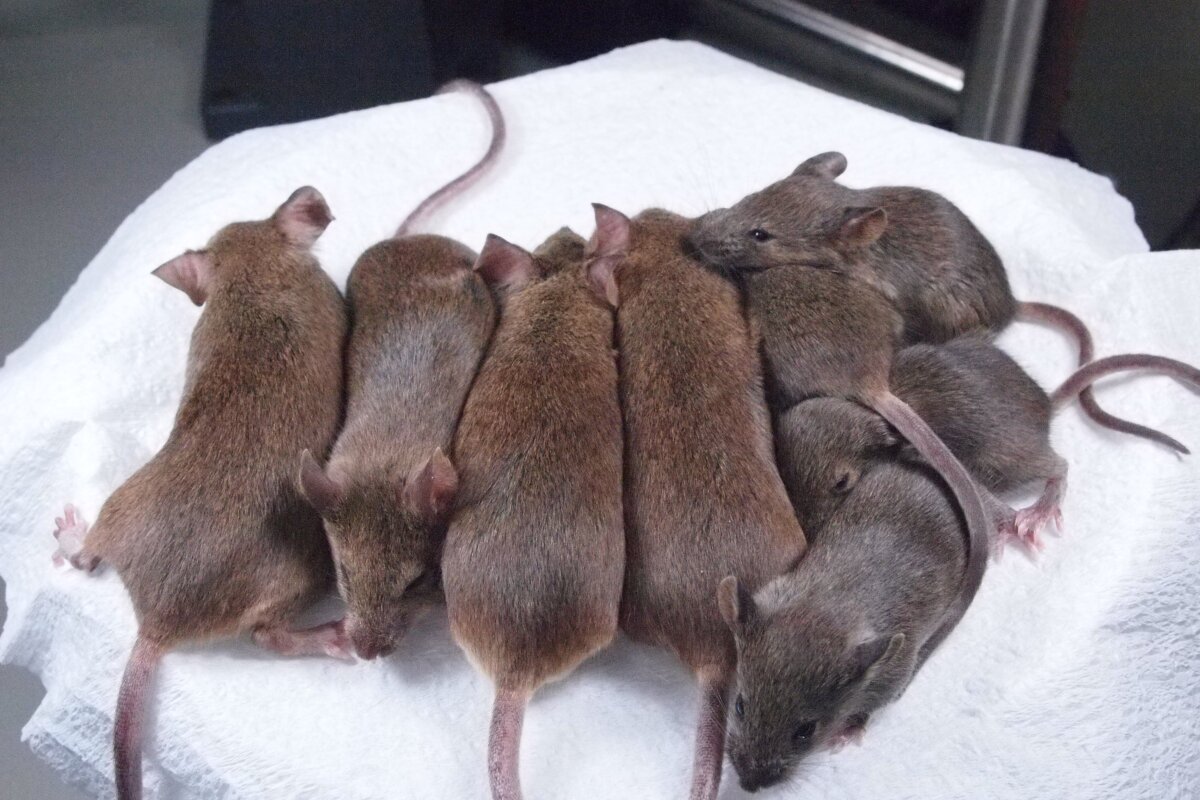
Using the technique that created Dolly the sheep, researchers from the RIKEN Center for Developmental Biology in Kobe, Japan, have identified a way to produce healthy mouse clones that live a normal lifespan and can be sequentially cloned indefinitely. In an experiment that started in 2005, the team led by Dr. Teruhiko Wakayama has used a technique called somatic cell nuclear transfer (SNCT) to produce 581 clones of one original "donor" mouse through 25 consecutive rounds of cloning.
Cloning of animals including humans has been a staple of science fiction at least since Aldous Huxley's Brave New World was published in 1932. Public interest reached a peak with the release of Steven Spielberg's Jurassic Park in 1993, but only became truly believable with the cloning of Dolly the sheep in 1996.

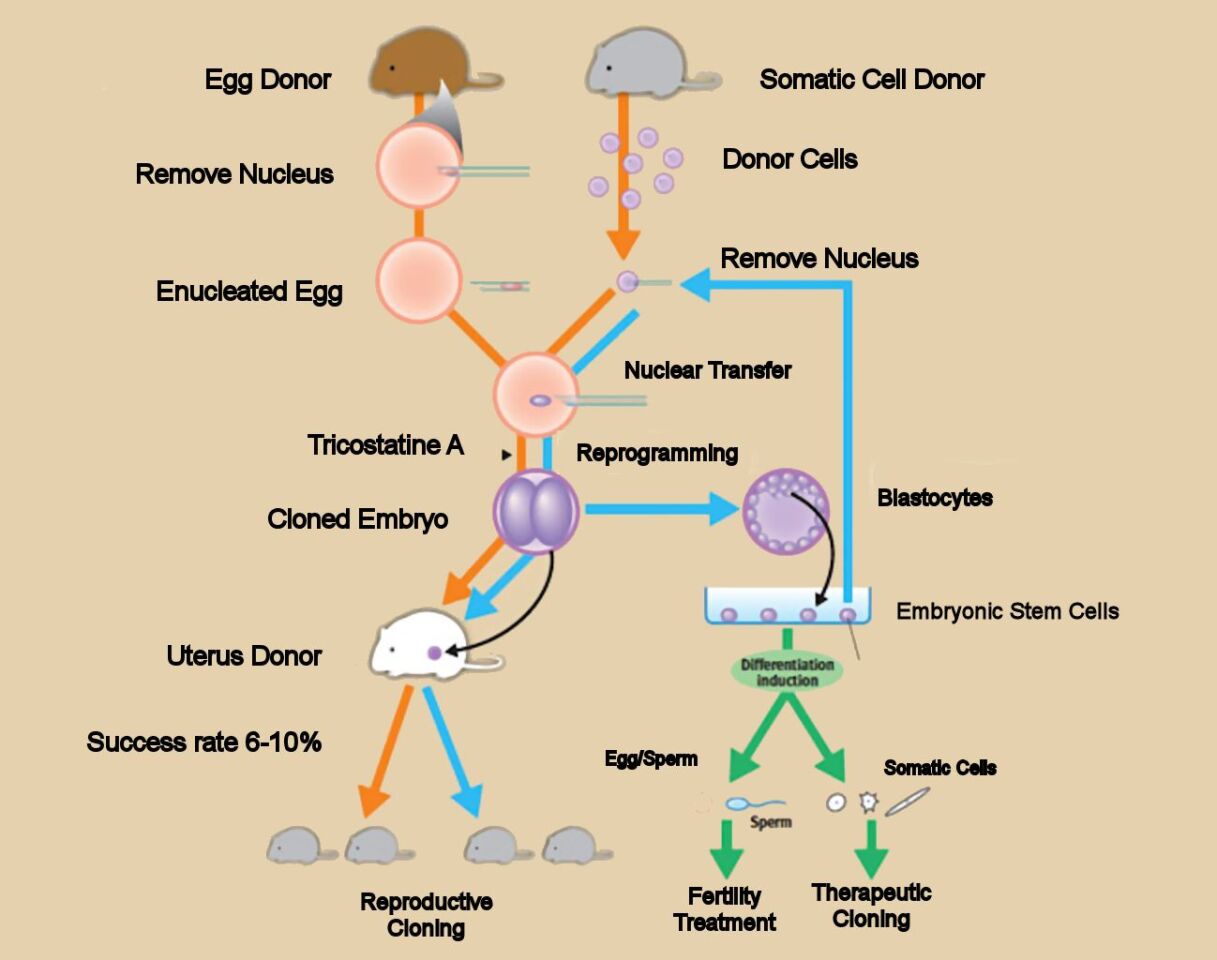
Dolly was cloned using the SCNT method, in which the nucleus of a somatic cell (any cell but egg or sperm) is removed and subsequently embedded in an egg cell whose nucleus had been previously removed. Once inside the egg cell, the somatic nucleus is reprogrammed by largely unknown factors to act as the nucleus of a fertilized egg, or zygote. At this point, a small electrical shock triggers the zygote to begin dividing. When a blastocyte has formed (an embryo containing about 100 eggs), the very young embryo is implanted into a suitable host animal, where it develops until birth.
SCNT has not been a terribly reliable procedure. To successfully clone Dolly required attempting SCNT with 277 eggs, of which one survived to adulthood. Even though the technique has been greatly improved in the past 20 years, the success rate in cloning mammals has hovered around 2 percent for many years. Even when cloning appeared to be successful, in that an adult animal was produced, some kind of genetic degradation was occurring. Rather like making a copy of a copy of a copy of a copy of an audio cassette, eventually further generations of clones became nonviable. They had accumulated too much genetic damage to survive.
Dr. Teruhiko Wakayama and his coworkers at Riken have greatly improved the cloning of mice. Their first advance was to use an SCNT method that applies trichostatin A (TSA), a histone deacetylase inhibitor, to the newly assembled cloned cells.
Histones are the packages for DNA, which, when they are acetylated, renders the DNA more accessible. The Riken group had observed that in the normal course of SCNT cloning, the histones in the somatic nucleus have been fully deacetylated, rendering the DNA difficult to access during cell division. To the contrary, in natural cells histone acetylization persists throughout the life of the cell. They hypothesized that TSA treatment would make the SCNT clone behave more like normal cells.
They were correct. The percentage of successful clones (successful meaning that the animal grew to adulthood) increased from about 2 percent to roughly 8-10 percent.
Serial cloning of animals is a sensitive test of genetic and epigenetic damage which can occur during cloning. In serial cloning, a clone is taken from the cloned animal. Each additional step is called a generation, so that a second generation clone is a clone of a clone.
When making mouse clones without TSA treatment, the Riken group found that the efficiency of serial cloning fell off with successive generations, to the extent that all seventh generation clones were nonviable. In contrast, when TSA treatment was included as part of the cloning process, they succeeded in producing many generations of clones, and the efficiency of successful cloning actually increased for the higher generations to about 12-14 percent. In addition, telomere length did not decrease through the serial cloning generations, indicating that, unlike Dolly, these mice were not born old. (Dolly had the same shortened telomere length as did her original, suggesting her cells were born with a genetic age of six years.)
The Riken lab has produced 581 cloned mice representing 25 generations of serial cloning produced from a single female mouse via the SCNT method including TSA treatment of the cloned cell. All the cloned mice had normal life spans and were physically and reproductively normal.
An important potential use for reproductive cloning of the sort carried out by Wakayama's group is in producing large numbers of particularly desirable animals for food production.
"If a 'super cow' that could produce a lot of milk or Kobe beef could be cloned at low cost, then not only consumers but also farmers would be happy," Dr. Wakayama told New Scientist.
The research was published this month in the journal Cell Stem Cell.