Most previous methods of producing methanol from carbon dioxide have involved lots of electricity, high pressures and high temperatures, and used toxic chemicals or rare earth elements like cadmium or tellurium. A team of researchers at the University of Texas at Arlington (UTA) has developed a new method they claim is safer, less expensive, and simpler than current approaches and can be scaled up to an industrial scale to allow some of the CO2 emitted from electrical power plants to be captured and converted into a useful fuel.
The simplest of the alcohol molecules (and poisonous to humans), methanol (CH30H) can be turned into a form of bio-diesel fuel and burned in engines. It is also an important chemical in the production of plastics, adhesives, and solvents.
In an interview with Gizmag, Dr Krishnan Rajeshwar, a distinguished professor of chemistry and biochemistry and co-founder of the Center for Renewable Energy, Science & Technology, CREST, at UT Arlington, described the new methanol production process developed by his team as a photo-electrochemical version of photosynthesis that occurs in plants.
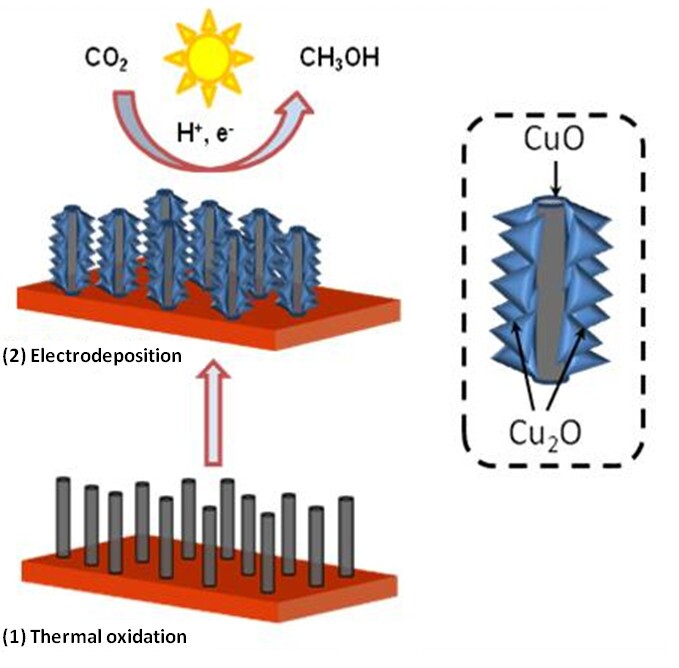
The heart of this technique uses a thermal process to coat copper oxide (CuO) nanowires with another form of copper oxide (Cu2O) and submerging them in a solution rich in carbon dioxide. The CuO-Cu2O hybrid nanorod arrays were then subjected to sunlight – or simulated sunlight in the lab – to trigger a chemical reaction and produce liquid methanol. The team says the experiments generated methanol with 95 percent electrochemical efficiency and avoided the excess energy input, also known as overpotential, of other methods.
When asked if this process might be used to create fuel for remote locations in Alaska and Canada, far from pipelines and roads, Dr. Rajeshwar thought it might be combined with the output of generators that make electricity, recovering the CO2 waste gas pollution to produce useful fuel.
The team's experiments were done on a very small scale, but the UTA team is now raising money to continue work on scaling up the process as part of their quest to create commercial products out of their research. “We hope the solutions in the lab are only the beginning”, said Carolyn Cason, VP for research at the University.
“As long as we are using fossil fuels, we’ll have the question of what to do with the carbon dioxide,” added Rajeshwar. “An attractive option would be to convert greenhouse gases to liquid fuel. That’s the value-added option.”
The UTA team, which also included Ghazaleh Ghadimkhani, Norma Tacconi, Wilaiwan Chanmanee, and Csaba Janaky, recently published their findings in the January 21, 2013 issue of Chemical and Engineering News.
Source: UT Arlington







