Sandy beaches are a delight for swimmers, surfers, sailors, and people strolling down the boardwalk. A horde of beautiful shells and buried coins (not to mention the occasional dropped ring) awaits the skilled beachcomber. Beach sand also carries within it a variety of traces of the history of that beach – a history which science can unlock. A prime example is the magnetic sands of Normandy.
Earle F. McBride and M. Dane Picard are geologists, the first at the University of Texas and the second at the University of Utah. Some time ago they visited Omaha Beach in Normandy primarily as tourists, but, being in the habit of collecting sand and sandstone samples wherever they travel, they collected samples of the Omaha Beach sand.
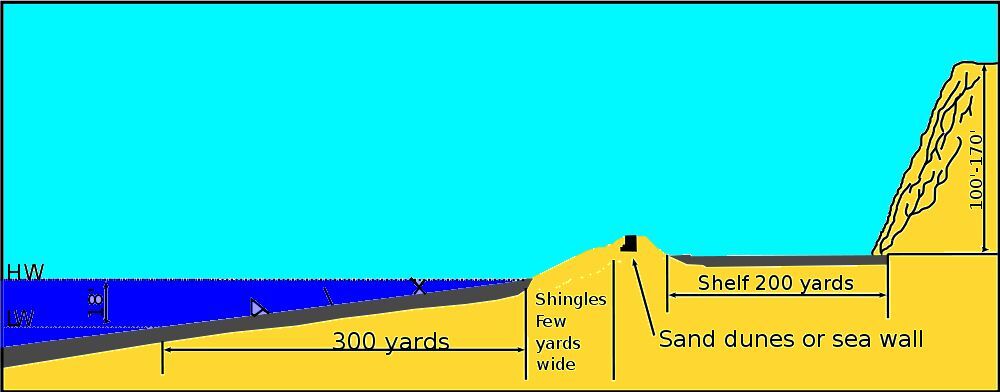
The beach itself is about 5 miles (8 km) long. The distance between the high water and low water marks is about 300 yards (275 m), reflecting a difference in coastal sea level of about 18 feet (5.5 m). At the high water mark there are a few yards covered with shingles (pebbles ranging up to about eight inches (20 cm) in diameter). Higher up, there are sand dunes in parts, and a sea wall in others, followed by a 200 yard (183 m) shelf of flat sand. The sand appears a pristine yellow-gray in color.
The two geologists examined the sand using several microscopes: a binocular optical microscope, a polarizing optical microscope and a scanning electron microscope, each of which provides different information on grain size, shape, roundness and composition. The sand is 78 percent degraded quartz, nine percent feldspar, four percent limestone and shell fragments, three percent heavy minerals, and two percent rock fragments. All in all, reasonably standard beach sand eroded from sedimentary rocks inland and carried to the shore by the Seine.

It is the remaining four percent that presented a bit of a mystery. The sand contained a large number of slightly rounded angular opaque grains that were magnetic. (Having only taken one sample, there is no way of knowing if four percent magnetic content is typical for Omaha Beach sand – samples from Utah Beach showed no signs of shrapnel.) Also in this magnetic fraction were small spherical beads of iron. Similar beads of glass also appeared in the larger part of the sample. McBride and Picard, after further testing, concluded that the magnetic grains, of which the sand contained four percent by weight, were indeed iron, accompanied by trace amounts of iron and glass beads.
How did all this shrapnel find its way to Omaha Beach? The most likely source is, of course, the Normandy landings. In the days following June 6, 1944, tens of thousands of tons of bombs and shells, mostly made of iron, exploded on the five beaches on which the Allied Powers were attempting to establish beachheads for a European invasion. The phrase "making the rubble dance" comes to mind, as that would have assisted in breaking up earlier pieces of shrapnel into smaller and smaller pieces.
The shrapnel grains range in size from about 0.002 to 0.04 inches (0.06 to 1.0 mm), with a mean grain size of about 0.01 inch (0.2 mm). They have a metallic appearance, with spots of red and orange rust and a good deal of corrosion. The grains appear to have retained their shape, but some of the sharp edges have become rounded by collisions with the surrounding sand. The grains show a laminated structure consistent with strong shock loading.
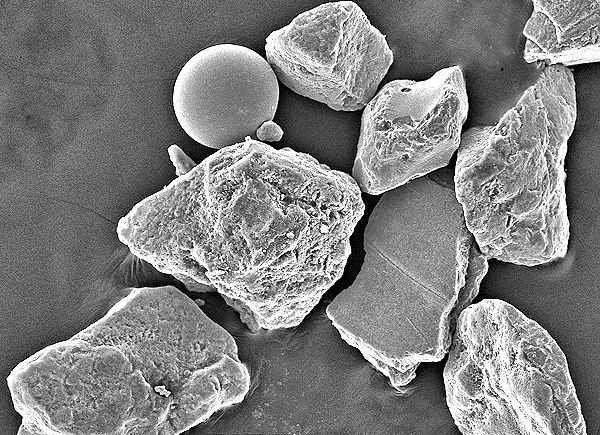
The more interesting question is what formed the iron and glass beads? In the geologist's sample were found 13 intact spherical iron beads, five hollow broken ones, and 12 glass beads. The iron beads are between 0.004 and 0.012 inches (0.1 to 0.3 mm) in diameter, are shiny in appearance, and appear nearly free of corrosion. The glass beads are spherical, have surprisingly smooth surfaces, and are nearly uniform in size at about 0.02 inches (0.5 mm) in diameter. The glass of which they are composed is clear with a certain amount of bubbles. Although one might expect the glass to mirror the composition of the beach sand, and be a silica glass, spectroscopic data reveals the presence of calcium, sodium, and magnesium as well.
McBride and Picard offer the following scenario for the formation of the beads – that the temperature of the explosions were hot enough, probably in excess of 2,200° F (1,200° C), to melt iron. It is known that bomb explosions often produce hollow metal beads. In the inverse of the process for forming ball bearings, molten iron is thrown up into the air, thereafter raining down as tiny spheres.
However, 2,200° F is not hot enough to thoroughly melt silica – this usually takes about 3,100° F (1,720° C). McBride and Picard postulate that the silica was heated along with sodium, calcium, and magnesium from the surrounding seawater – these are the three most abundant elements in seawater save for chlorine. Reacting with the silica grains in the sand, probably through ion-exchange reactions, the result would be formation of a glass that is primarily silica, but which has a substantially depressed melting point.
Also aiding such a glass-forming reaction is that exposure to a shock wave, such as that produced by an explosion, is well-known to greatly increase the rate of chemical reactions from the rate expected at a particular temperature. After this, one is again forming glass ball bearings through the same process that formed the iron spheres.
McBride and Picard believe that after a century or so, the combined effects of corrosion and abrasion will leach the sand at Omaha Beach of the evidence that a great battle was fought there.
- And on the pedestal these words appear:
"My name is Ozymandias, king of kings:
Look on my works, ye Mighty, and despair!"
Nothing beside remains. Round the decay
Of that colossal wreck, boundless and bare
The lone and level sands stretch far away."
Percy Bysshe Shelley, Ozymandias
Source: University of Texas







